
Collagen Peptides: The Key to Aging Gracefully
Chris D. Meletis, N.D. Collagen is the most abundant protein in the body, and it’s the building block of connective tissues like skin, muscles, bone, tendons, and ligaments. You need collagen for y...

Small Changes, Big Impact: Easy Daily Habits for Optimal Health
“We are what we repeatedly do. Excellence, then, is not an act, but a habit.” – Aristotle Embarking on a journey towards better health and well-being doesn't always require grand gestures or life-a...

Thanksgiving Detox: Supplements to Help You Recover After the Big Day
As the vibrant leaves of fall give way to the cozy charm of Thanksgiving, it's hard not to get swept up in the spirit of feasting and family. This cherished holiday often brings with it a bounty o...
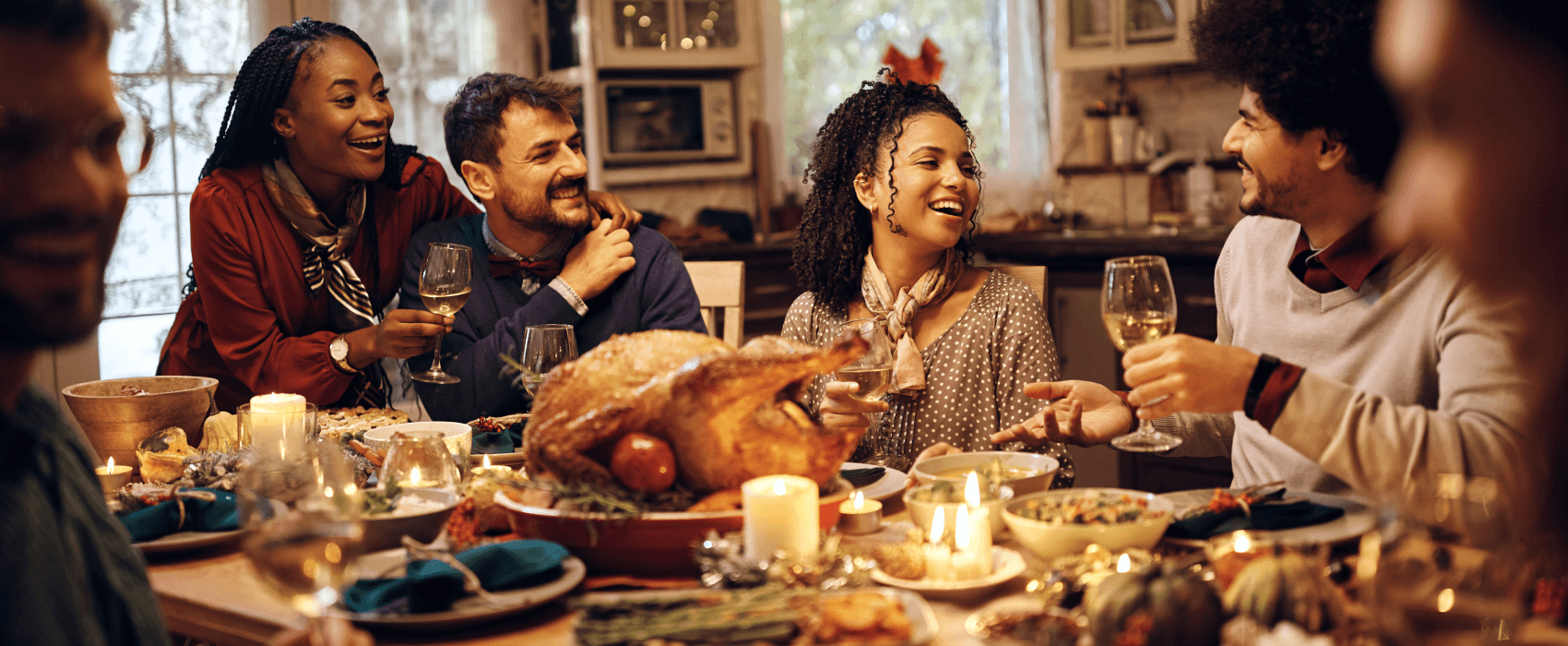
The Surprising Nutrition in Thanksgiving Classics
As the leaves turn golden and the air carries a crisp whisper of autumn, our thoughts inevitably drift to the warmth of Thanksgiving—a time for gratitude, family, and of course, a bountiful feast. ...

Your 2023 Gift Guide to Better Health
As the holiday jingles start to play and the festive cheer envelops us, we’re reminded that the best gifts are those that nurture health and vitality. This guide is perfectly timed for the holiday ...

Don't Let Halloween Candy Deplete Your Magnesium Levels
Halloween is just around the corner, and that means spooky costumes, jack-o'-lanterns, and of course, an abundance of sweet treats. For health-conscious moms, it can be a real challenge to balance ...
Why Fall is the Best Time to Strengthen Your Immunity
As the leaves begin to change and the crisp air of autumn surrounds us, there's a natural inclination to cozy up, sip on warm beverages, and prepare for the colder months ahead. But beyond the beau...
Is Magnesium Glycinate the Best Form of Magnesium for You?
Chris D. Meletis, N.D. Magnesium is an essential mineral that has many important roles in health. It can support healthy blood pressure, keep bones healthy, improve mood and feelings of well-being,...

It's Non-GMO Month: Here's What You Need To Know
Celebrate Non-GMO Month The month of October is Non-GMO Month, let's explore the world of non-GMO foods and supplements and understand why they are essential for our health and the environment. The...

